Choosing a dietary supplement can feel overwhelming when nearly 82% of weight-loss supplements are mislabeled, leaving you uncertain about what you’re actually consuming. This widespread problem of inaccurate labeling and hidden ingredients puts your health at risk and makes it nearly impossible to verify if a product delivers on its promises. Understanding supplement transparency, the practice of clearly disclosing all ingredients, dosages, sourcing, and testing results, empowers you to make safer, more informed choices. This guide explains what transparency means in the supplement industry, why it’s essential for your health, and how to identify products that prioritize honest disclosure over marketing hype.
Table of Contents
-
Why transparency matters: scientific backing and consumer trust
-
How to evaluate supplement transparency when choosing products
-
Explore trusted supplement options with verified transparency
Key Takeaways
Point Details
What transparency means Transparency means clear disclosure of every ingredient exact dosages sourcing and third party testing results so you can verify safety and efficacy.
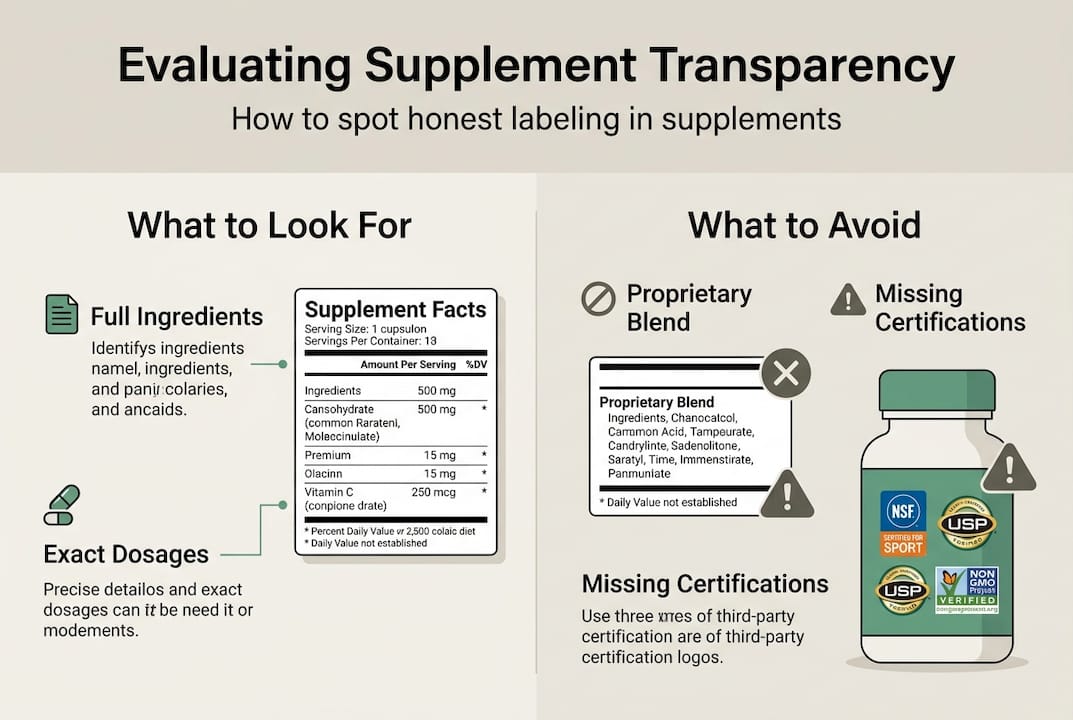
Label content essentials Complete ingredient lists with common and scientific names exact dosages for each ingredient clear sourcing information and allergen disclosures.
Avoid proprietary blends Proprietary blends conceal individual amounts making it impossible to confirm effective dosing and should be avoided.
Verify with certifications Rely on third party testing certificates facility information and clear testing methods to confirm the label matches the product.
What is supplement transparency?
Supplement transparency refers to clear disclosure of all ingredients, their exact dosages, sourcing information, and third-party testing results on dietary supplement labels. This practice enables you to verify product quality, safety, and efficacy before making a purchase. When a manufacturer commits to transparency, they provide complete information rather than hiding behind vague claims or proprietary formulas.
Transparent labeling allows you to cross-reference ingredients with published research studies to confirm whether the product contains clinically effective amounts. For example, if you’re considering immune support supplements, transparency lets you verify that vitamin C or zinc appears in amounts shown to benefit immune function in peer-reviewed trials. Without this information, you’re essentially gambling with your health and money.
Key components of transparent supplement labels include:
-
Complete ingredient lists with both common and scientific names
-
Exact dosage amounts for every active ingredient, not ranges or estimates
-
Clear identification of ingredient sources and origins
-
Third-party testing certificates confirming label accuracy
-
Manufacturing facility information and quality control processes
-
Allergen warnings and potential contaminant disclosures
Transparency contrasts sharply with proprietary blends, which list multiple ingredients under a single total weight without revealing individual amounts. This practice prevents you from knowing whether each ingredient appears in therapeutic doses or merely trace amounts included for label appeal. A truly transparent product breaks down every component so you can make educated decisions based on science rather than marketing promises.
Manufacturers who embrace transparency demonstrate confidence in their formulations and respect for consumer intelligence. They understand that informed customers become loyal customers when products deliver measurable results backed by honest disclosure.
Regulations and realities of supplement labeling accuracy
The FDA establishes Current Good Manufacturing Practices under 21 CFR Part 111, requiring manufacturers to follow specific procedures including visual examination of components, quarantine until testing, lot identification, and proper handling of rejected products to ensure label accuracy. These regulations aim to protect you from contaminated, mislabeled, or substandard products entering the market.
Despite these regulatory requirements, the reality of supplement labeling reveals troubling discrepancies:
Issue Rate Impact
Weight-loss supplements mislabeled 82% Consumers receive unknown or incorrect ingredient amounts
Saw palmetto products meeting USP standards 1 in 28 Vast majority fail basic quality benchmarks for fatty acid content
Weight-loss products adulterated with pharmaceuticals 37.5% Dangerous undisclosed drugs like sibutramine pose serious health risks
These label inaccuracy statistics expose a significant disconnect between what regulations require and what actually reaches store shelves. The FDA lacks resources for comprehensive pre-market testing, relying instead on post-market surveillance and consumer complaints to identify problems. This reactive approach leaves you vulnerable to products that may contain less than claimed amounts, entirely different ingredients, or dangerous contaminants.
Inaccurate or incomplete labeling creates several risks for you:
-
Inability to achieve desired health outcomes due to underdosed active ingredients
-
Unexpected interactions with medications when unlisted ingredients are present
-
Allergic reactions from undisclosed allergens or fillers
-
Toxicity from contaminated products or pharmaceutical adulterants
-
Wasted money on ineffective formulations masquerading as legitimate supplements
The supplement safety landscape continues evolving as regulators and consumer advocates push for stronger enforcement. Meanwhile, products like Nutrigo Lab Strength that prioritize verified transparency stand out by voluntarily exceeding minimum regulatory standards.
Understanding this regulatory reality helps you recognize why personal due diligence remains essential when selecting supplements, even from seemingly reputable brands.
Why transparency matters: scientific backing and consumer trust
Scientific backing requires clinically effective doses, but proprietary blends hinder verification by hiding individual ingredient amounts. When you can’t confirm dosages, you can’t cross-reference products with randomized controlled trials that establish therapeutic thresholds. Full disclosure allows you to match supplement contents against peer-reviewed research to verify whether formulations contain amounts proven effective.
Consider creatine monohydrate, extensively studied for muscle building. Research consistently shows 3 to 5 grams daily produces measurable strength gains. A transparent label states exactly how much creatine each serving provides, letting you confirm the product delivers research-backed amounts. A proprietary blend listing creatine alongside ten other ingredients under a 2000mg total weight leaves you guessing whether you’re getting 1500mg or just 50mg.
The controversy surrounding proprietary blends centers on competing interests:
-
Industry perspective: Manufacturers claim proprietary formulas protect intellectual property and prevent competitors from copying successful formulations
-
Consumer advocate position: Critics argue proprietary blends primarily protect profit margins by enabling underdosing of expensive ingredients while maintaining premium pricing
-
Regulatory debate: Proposed legislation like the Dietary Supplement Listing Act would require FDA listing of all ingredients with specific amounts, eliminating proprietary blend loopholes
Retailers increasingly enforce clean label standards by banning products with certain additives, responding to consumer demand for simpler, more transparent formulations. This market-driven pressure complements regulatory efforts to improve disclosure practices across the industry.
Transparency builds consumer trust by demonstrating manufacturer confidence and accountability. When a company willingly subjects products to third-party testing and publishes detailed certificates of analysis, they signal commitment to quality that goes beyond minimum legal requirements. This openness creates a relationship based on mutual respect rather than information asymmetry.

Research on consumer engagement with transparency reveals an interesting paradox: people demand transparency but often don’t deeply engage with disclosed information. However, the mere availability of detailed data increases trust and purchase intent, even among consumers who don’t thoroughly analyze every ingredient. This suggests transparency serves both practical verification purposes and symbolic trust-building functions.
Pro Tip: Before purchasing any supplement, search for peer-reviewed studies on its key ingredients using PubMed or Google Scholar. Verify that the product contains amounts matching those shown effective in research. Avoid any product hiding ingredient quantities behind proprietary blend labels, as this prevents meaningful comparison with scientific literature.
The connection between exercise supplements and synergy further illustrates why transparency matters. When combining multiple supplements, you need precise dosage information to avoid excessive intake or negative interactions. Transparent labeling enables you to calculate total daily intake across all products, maintaining safe and effective supplementation strategies.
How to evaluate supplement transparency when choosing products
Evaluating supplement transparency requires systematic examination of several label features and external verification sources. Start by checking these essential transparency indicators:
-
Complete ingredient list: Every active and inactive ingredient should appear with both common and scientific names
-
Exact dosage amounts: Milligrams or micrograms for each ingredient, not combined totals or ranges
-
Third-party certification seals: NSF International, USP Verified, or ConsumerLab.com logos indicating independent testing
-
Certificates of analysis (COA): Accessible documents showing batch-specific testing results for purity and potency
-
Manufacturing information: Facility location, GMP certification status, and contact details for questions
-
Clear allergen statements: Explicit disclosure of common allergens and potential cross-contamination risks
Transparency indicator What it means Why it matters
Individual ingredient dosages Each component listed with specific amount Allows verification against clinical research and prevents underdosing
NSF or USP certification seal Independent lab verified contents match label Confirms third-party validation rather than manufacturer self-testing
Accessible COA Batch-specific purity and potency test results Provides transparency into actual product quality beyond label claims
Source identification Origin of raw materials disclosed Enables assessment of ingredient quality and ethical sourcing
No proprietary blends All ingredients individually quantified Eliminates information hiding and enables informed decision-making
Recognize these red flags indicating poor transparency:
-
Proprietary blends under 1500mg total weight, suggesting minimal amounts of listed ingredients
-
Absence of third-party certification despite premium pricing
-
Unavailable or refused certificates of analysis when requested
-
Vague terms like “herbal blend” or “natural complex” without specifics
-
Dramatic marketing claims unsupported by disclosed ingredient amounts
-
Missing contact information or unresponsive customer service
Pro Tip: Use supplements with NSF or USP seals as your transparency benchmark. These certifications require rigorous testing and ongoing audits, setting a quality standard against which you can compare other products. When a supplement lacks certification, it should compensate with exceptionally detailed labeling and readily available COAs.
Practical research shows that avoiding proprietary blends under 1500mg and seeking products with certificates of analysis significantly improves your chances of getting accurately labeled supplements. The study also found that structured ad presentations emphasizing specific ingredients and amounts boost consumer trust more effectively than holistic marketing approaches focusing on general wellness claims.
When evaluating eye health supplements or digestive products like Fibre Select, apply these same transparency standards. Look for specific amounts of lutein, zeaxanthin, or fiber rather than vague “eye support blend” or “digestive health complex” labels. The more precisely a manufacturer discloses contents, the more confidence you can have in product quality.
Trust detailed, structured labels over emotionally appealing but vague marketing language. A supplement claiming to “revolutionize your health” without specifying ingredient amounts should raise immediate concerns. Conversely, a product listing every ingredient with exact dosages, sources, and testing certifications demonstrates the transparency necessary for informed decision-making.
Remember that transparency extends beyond the label itself. Reputable manufacturers maintain informative websites, responsive customer service, and willingness to answer detailed questions about sourcing, manufacturing, and testing. This accessibility signals organizational commitment to transparency as a core value rather than a marketing tactic.
Explore trusted supplement options with verified transparency
Navigating the supplement market becomes significantly easier when you have access to expert reviews that prioritize transparency and ingredient verification. RankOfSupplements specializes in evaluating products based on scientific evidence, label accuracy, and manufacturer transparency rather than marketing hype. Our detailed analyses help you identify supplements that deliver on their promises with honest disclosure.
Explore our comprehensive rankings for immune support supplements that clearly disclose vitamin and mineral amounts backed by clinical research. Our review of Nutrigo Lab Strength examines ingredient transparency and dosing accuracy to help you make informed choices about performance supplements. For digestive health, our Fibre Select reviews analyze fiber content disclosure and quality verification. Each review provides the transparency information you need to select supplements confidently.
Frequently asked questions
What is supplement transparency?
Supplement transparency means manufacturers clearly disclose all ingredients, their exact dosages, sourcing information, and third-party testing results on product labels. This practice allows you to verify quality, safety, and effectiveness before purchasing. Transparent companies provide complete information rather than hiding behind vague claims, proprietary formulas, or minimal disclosure.
How does supplement transparency benefit consumers?
Transparency enables you to verify that products contain clinically effective amounts of active ingredients by cross-referencing labels with peer-reviewed research. It helps you avoid allergens, identify potential medication interactions, and make cost-effective choices based on actual ingredient value. Transparency also builds trust between manufacturers and consumers by demonstrating accountability and quality commitment beyond minimum legal requirements.
What are FDA regulations for supplement labeling?
The FDA requires manufacturers to follow Current Good Manufacturing Practices under 21 CFR Part 111, including component testing, lot identification, and proper handling of rejected materials. However, supplements don’t require pre-market approval like pharmaceuticals. The FDA relies on post-market surveillance to identify problems, meaning products can reach consumers before testing reveals inaccuracies or safety issues.
How can I quickly recognize a transparent supplement?
Look for products listing every ingredient with specific dosage amounts, third-party certification seals like NSF or USP, and accessible certificates of analysis showing batch-specific testing results. Avoid supplements using proprietary blends that hide individual ingredient quantities. Transparent manufacturers also provide detailed sourcing information and maintain responsive customer service willing to answer technical questions about formulations.
How do proprietary blends affect supplement transparency?
Proprietary blends list multiple ingredients under a single combined weight without revealing individual amounts, preventing you from verifying whether each component appears in clinically effective doses. This practice eliminates transparency by making it impossible to compare products against scientific research or calculate total daily intake when using multiple supplements. Critics argue proprietary blends primarily protect profit margins by enabling underdosing of expensive ingredients while maintaining premium pricing.
What certifications indicate a transparent supplement?
NSF International and USP Verified certifications confirm that independent laboratories have tested products and verified contents match label claims. These seals require ongoing audits and batch testing, providing stronger assurance than manufacturer self-testing. Certificates of analysis (COA) offer batch-specific data on purity and potency, giving you transparency into actual product quality beyond standard label information.
Why are supplement labels sometimes inaccurate?
Regulations allow some variation in dosages, and testing or quality control lapses occur during manufacturing. Economic incentives may encourage underdosing expensive ingredients or hiding pharmaceutical adulterants to enhance perceived effectiveness. The FDA’s reactive oversight approach means problems often aren’t discovered until after products reach consumers, allowing inaccurate labels to persist in the market.
